1. Donate Plasma: Earn Money. Save Lives. | Octapharma Plasma
New Donors · OctaApp · OctaPass · About Plasma
You can help create life-changing medicines & earn money when you donate plasma. Be a real hero; donate plasma!

2. Plasma - Octapharma
Collecting plasma in the USA. In the USA, Octapharma Plasma, Inc. collects, tests and supplies human blood plasma for manufacture into life-saving therapies.
Our life-savings therapies are made from human plasma. 80% of our plasma comes from company-owned donation centres.

3. New Donors | Octapharma Plasma
Find out everything you need to know about plasma donor eligibility, first time plasma donation requirements and new donor bonus payment incentives.

4. Acta Pharmaceutica - Sciendo
Acta Pharmaceutica (AP) is an international, scientific, multidisciplinary journal, devoted to the pharmaceutical and allied sciences, which contains ...
Acta Pharmaceutica (AP) is an international, scientific, multidisciplinary journal, devoted to the pharmaceutical and allied sciences, which contains articles predominantly on core biomedical and...

5. Acta Scientific | Open Access Journals Publishing Group - Acta Scientific
17 mei 2024 · Acta Scientific Pharmaceutical Sciences (ASPS)(ISSN: 2581-5423) ... The plasma concentration may fluctuate as a result of this type of ...
Osmotic Drug Delivery System - A Review

6. ACTA Pharmaceutica Sciencia
Bevat niet: plasma | Resultaten tonen met:plasma
As of 01.10.2023 , the print version has been stopped and the journal continues as an electronic version.
7. [PDF] Quantification of Cyclobenzaprine in Human Plasma by LC/MS/MS and ...
30 okt 2020 · Acta Scientific Pharmaceutical Sciences (ISSN: 2581-5423). Volume 4 Issue 11 November 2020. Quantification of Cyclobenzaprine in Human Plasma ...
8. [PDF] Determination of Topiramate in Human Plasma using Liquid ...
The LC-MS/MS method for determination of the anti-epileptic drug topiramate (TPM) in human plasma was developed and validated for pharmaco*kinetic and ...
9. Plasma-derived medicines: access and usage issues - PMC - NCBI
Unlike the situation for most pharmaceuticals, the costliest component of the manufacture of plasma protein therapies is the raw material, plasma. In well- ...
As a library, NLM provides access to scientific literature. Inclusion in an NLM database does not imply endorsem*nt of, or agreement with, the contents by NLM or the National Institutes of Health. Learn more: PMC Disclaimer | PMC Copyright Notice

10. [PDF] ACTA PHARMACEUTICA SCIENCIA - Medipol Üniversitesi
Representative chromatogram of blank plasma (A), fexofenadine pure drug (B),. Figure 4. Representative chromatogram of blank plasma (A), fexofenadine pure drug ...
11. Acta Pharmaceutica | A quarterly pharmaceutical journal
Bevat niet: plasma | Resultaten tonen met:plasma
A quarterly journal of Croatian Pharmaceutical Society and Slovenian Pharmaceutical Society, dealing with all branches of pharmacy and allied sciences.
12. Structure‒tissue exposure/selectivity relationship (STR) correlates ... - NCBI
Acta Pharm Sin B. 2022 May; 12(5): 2462–2478. Published online 2022 Feb 23. doi: 10.1016/j.apsb.2022.02.015 ... In drug development process, drug exposure in plasma (drug concentration and area under the ...
Drug optimization, which improves drug potency/specificity by structure‒activity relationship (SAR) and drug-like properties, is rigorously performed to select drug candidates for clinical trials. However, the current drug optimization may overlook ...

13. [PDF] Clinical Relevance of High Plasma Trough Levels of the ... - UMC Utrecht
21 mrt 2024 · last drug intake and plasma sampling date and time were used to ... Clin Chim Acta. 2017;472:26–29. 15. Balakirouchenane D, Guégan S ...
14. A novel HPLC method for selexipag in human plasma and application to a ...
23 nov 2023 · ... Acta Chromatographica. Volume/Issue: Accepted Manuscript ... Preparation of drug solution and plasma. The stock standard solution ...
Abstract A novel simple and cost effective HPLC technique was presented for the quantification of selexipag (SLP) in human plasma sample and the technique's applicability to a pharmaco*kinetic investigation. Chromatographic separation was achieved with C18 (5 µm × 4.6 mm × 150 mm) column, at 30 °C with isocratic elution, mobile phase composed of solution A (acetonitrile), and solution B (0.5% formic acid) (65:35 v/v) at flow rate 1.2 mL min−1. The linearity range is 10–150 ng mL−1. As sample preparation step human plasma was precipitated with acetonitrile and the detection was provided at 300 nm. The retention time is 8.20 ± 0.02 min. LOD is found to be 3.3 ng mL−1 for drug. The method was applied to the analysis of SLP in human plasma with good recovery as 97.83%. Validation of the studied methods was carried out according to EMA guideline. The new method applied on a prototype pharmaco*kinetic study by administration of 800 μg SLP to a healthy volunteer and parameters like AUC0–24, AUC0–∞, Cmax, tmax, and t1/2 were assessed.

15. [PDF] ACTA POLONIAE - Polskie Towarzystwo Farmaceutyczne
Acta Poloniae Pharmaceutica с Drug Research, Vol. 71 No. 2 pp. 215с221, 2014 ... tein component of blood plasma and due to its endogenous and exogenous ...
16. Biomedical and Pharmaceutical Applications of Inductively Coupled ...
Determination of heavy metals in selected drug substances by inductively coupled plasma-mass spectrometry. ... Analytica Chimica Acta. 2002; 453 :311-323 ...
Medical science is a field of study that is relevant to all people, but the development of pharmaceutical, biomedical and life science is of particular importance. In these fields, further studies are being established to determine with incredible accuracy the quantities and concentration of inorganic elements and organic compounds, such as nucleotides, sulphur and phosphorous containing peptides and proteins, to be used in all kinds of drugs. Since 1980, inductively coupled plasma-mass spectrometry (ICP-MS) has emerged as a new and powerful technique for elemental and isotopic analysis. It provides a means for the analysis of an extremely wide range of elements and the co-analysis of most elements in the periodic table. It can also be used for qualitative, quantitative and semiquantitative analysis and for the measurement of isotopic ratios through mass-to-charge ratios. In recent years, ICP-MS has emerged as the best technique for the quantification of inorganic impurities in pharmaceutical and biomedical applications. This chapter focuses on introducing the applications of ICP-MS in the pharmaceutical and biomedical fields. Some problems facing ICP-MS are also presented at the end of this chapter.

17. Chronic angiotensin (1-7) injection accelerates STZ-induced diabetic ...
Acta Pharmacol Sin 2008 Jul; 29 (7): 829–837. ©2008 CPS and SIMM. Full ... Plasma and renal Ang II levels were measured by radioimmunoassay. Ang-con ...
18. A sensitive, heart-cutting, two-dimensional - AKJournals
22 nov 2022 · Drug-free plasma samples from twelve donors were assayed. ... Acta 2021, 1184, 339022. 11. Pugajeva, I.; Ikkere, L. E.; Jansons, M ...
19. [PDF] ACTA PHARMACEUTICA SCIENCIA - Medipol Üniversitesi
the drug concentration (1/X2). RESULTS AND DISCUSSION. It was reported earlier that abiraterone is unstable in blood/plasma in the ab- sence of fluoride 14 ...
20. Academisch Centrum Tandheelkunde Amsterdam
Bevat niet: pharma plasma
Het Academisch Centrum Tandheelkunde Amsterdam (ACTA) leidt tandartsen op om het gebit van mensen te verzorgen en doet onderzoek om die zorg te verbeteren.

21. Influence d'acides gras saturés et non saturés sur la pression de filtration ...
4 mrt 2009 · Section title: Paper. Acta Haematol 1973;49:306–313 (DOI:10.1159 ... Thus, with the technique of filtration pressure of platelet-rich plasma ...
Abstract. The influence of the salts of saturated and non-saturated fatty acids from C8 to C22 on the aggregation of human platelets is compared within the framework of 2 different techniques: the test of filtration pressure, in correlation with the classical photometric test of aggregation. Thus, with the technique of filtration pressure of platelet-rich plasma, one finds an increase in filtration pressure as a function of the number of carbon molecules in the chain used. The presence of a double bond generally results in a decrease in filtration pressure. In the classical photometric test the fatty acids induce a progressive and irreversible deviation curve, demonstrating both the aggregation of platelets and the probable bonding of the fatty acids with plasmatic proteins. The presence and intensity of platelet aggregation are related to the molecular weight of the chain of fatty acids under consideration and inhibited by the presence of double bonds. From the similarity of the results obtained with the 2 techniques, one might surmise that the filtration pressure test is a new approach to platelet aggregation.

22. Ecofriendly micellar mediated spectrofluorimetric method for ...
16 jul 2024 · ... drug safinamide in pharmaceutical formulation and spiked human plasma ... Acta Part A Mol. Biomol. Spectrosc. 244, 1–7 (2021). Article ...
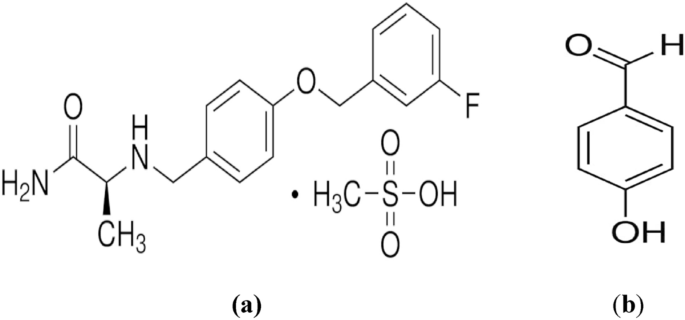
A novel, highly sensitive and eco-friendly micellar-mediated spectrofluorimetric method was developed and validated for the determination of the novel antiparkinsonian drug safinamide mesylate in the presence of its related precursor impurity, 4-hydroxybenzaldehyde. The proposed approach relies on increasing the inherent fluorescence emission at 296 nm of safinamide, by forming hydrogen bonds between the mentioned drug and sodium dodecyl sulfate in the micellar system using 0.1 N HCl as a solvent, following excitation at 226 nm. A thorough investigation was conducted into the experimental factors affecting spectrofluorimetric behavior of the studied drug. A linearity plot of safinamide over the concentration range of 10.0–1000.0 ng/mL against the relative fluorescence intensities was established. The proposed method demonstrated excellent sensitivity down to the nano-gram level with detection and quantitation limits of 1.91 and 5.79 ng/mL, respectively. The studied drug was effectively determined in Parkimedine® Tablets. Furthermore, the proposed method allows for ultrasensitive quantification of safinamide in spiked human plasma, with satisfactory percentage recovery (98.97–102.28%). Additionally, the greenness assessment using the advanced green certificate classification approach, the complementary green analytical procedure index (Complex-GAPI), and the analytical GREEness metric approach (AGREE), along with the practicality check using the Blue Applicability Grade Index...
23. [PDF] Bioequivalence Study of Two Different Formulations of Ceftiofur Following ...
The chromatogram acquired from an extract of the drug-free plasma sample is presented in ... Analytica Chimica Acta, 512(1), 75–84. [CrossRef]. El-Gendy ...
24. [PDF] Cathodic Stripping Voltammetric Determination of Febuxostat in ...
Portugaliae Electrochimica Acta 2016, 34(5), 343-353. DOI: 10.4152/pea ... Drug-free plasma samples were obtained from healthy people, and stored frozen.